Blood Culture Accuracy is Vital for Patient Care Quality and Requires Following Evidence-Based Best Practices to Achieve Optimal Fill Volumes.
In 2024, hospitals were impacted by an unprecedented shortage of blood culture media bottles.1 In response, healthcare providers employed conservation measures that included changes to blood culture collection practices—such as drawing single-set blood cultures—that may have deviated from established best practices.
As the supply of blood culture bottles has gradually recovered to pre-shortage levels, it’s time to review blood culture collection protocols and practices to ensure that they are aligned with recommended clinical practices.2 Returning to evidence-based, blood culture collection best practices is not just about compliance— it’s essential for improving patient care quality, outcomes, and hospital performance.
Blood Culture Volume is Crucial to Pathogen Recovery
In fact, the yield of pathogens increases in direct proportion to the volume of blood cultured, as portrayed in the figure below measuring the cumulative sensitivity of blood culture sets.3,4
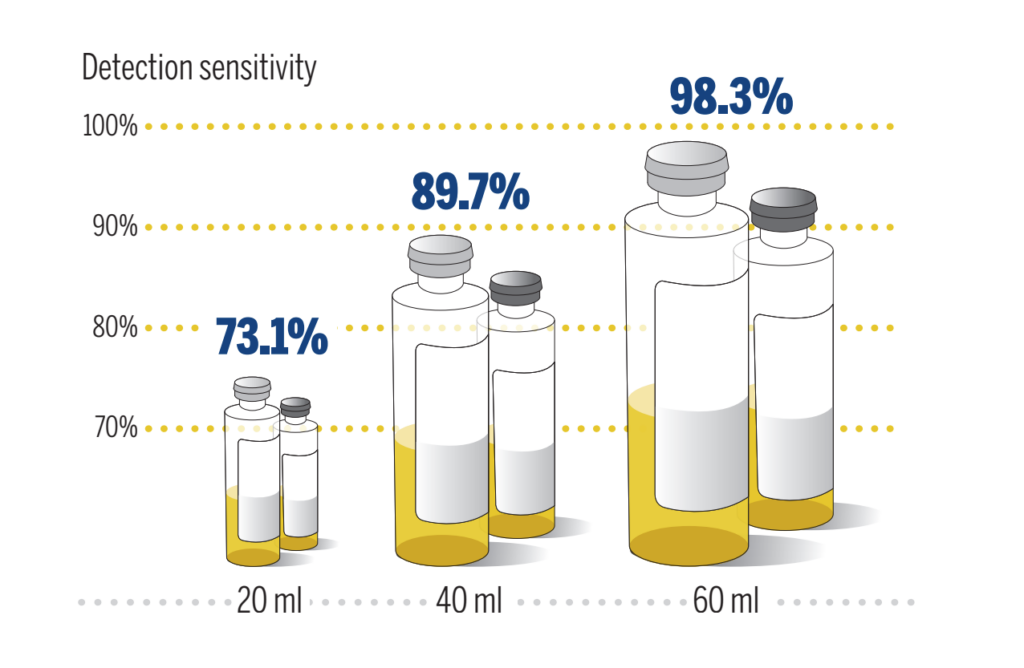 Adapted from Lee A, Mirrett S, Reller LB, Weinstein MP. Detection of Bloodstream Infections in Adults: How Many Blood Cultures Are Needed? J Clin microbiol 2007y;45:3546-3548
Adapted from Lee A, Mirrett S, Reller LB, Weinstein MP. Detection of Bloodstream Infections in Adults: How Many Blood Cultures Are Needed? J Clin microbiol 2007y;45:3546-3548
Detecting potential pathogens requires multiple blood culture sets to be drawn within hours of each other; an adult blood culture should contain at least 2 blood culture sets that equates to approximately 40 mL of blood volume. If an inadequate blood volume is collected, it becomes more challenging to reliably recover pathogens within the sample. 5,6
Unfortunately, an insufficient fill volume may lead to a false-negative result for the adult patient. A misdiagnosis, like a false negative, can cause treatment delays and an increased risk of patients developing—or dying from—bloodstream infections.
Mitigating False-Positive Results due to Contamination Events and the National Patient Safety Measure
In addition to the risk of a false-negative event, a false-positive result due to contamination may also severely impact care quality. Recently, a representative from the Centers for Disease Control and Prevention (CDC) called blood culture contamination (BCC) a “patient safety event.”7,8 Given its dire consequences, the CDC proposed BCC rate as the first clinical laboratory-based national patient safety measure when it was endorsed in December 2022.9 Overall BCC rates for hospitals should not exceed 3%, per the American Society for Microbiology (ASM) and the Clinical Laboratory Standards Institute (CLSI).10,11 A goal of 1% is possible when best practices are followed and should be the target for facilities, according to CLSI and the CDC.5,11
Calculating the blood culture contamination rate includes dividing the total number of contaminated blood culture sets by the total number of ELIGIBLE blood culture sets collected during the evaluation period.

For example, if an institution has 200 blood culture sets drawn per month on 100 patients (each patient having 2 sets drawn within 24 hours), and one set grows Staphylococcus epidermidis while the other set has no growth, then the institution’s contamination rate is 0.5%.
Contamination Rate 1/200= 0.5%.
Two sets are required to determine if a blood culture is a contamination or a true blood stream infection. If only a single set is drawn within a 24-hour period, the blood culture result is compromised as the volume may be insufficient for accurate testing. Furthermore, single-set blood cultures should be excluded in blood culture contamination rate calculations.12
If a patient only has one set drawn, and that set grows out a skin commensal organism, it is difficult to make a determination as there is no second set to confirm or refute a BSI with a commensal organism. Single-set blood cultures, a temporary measure during the blood culture bottle shortage, can result in inaccurate test results, and should only be implemented as a last resort measure.1
Hospital Accountability and Diagnostic Stewardship
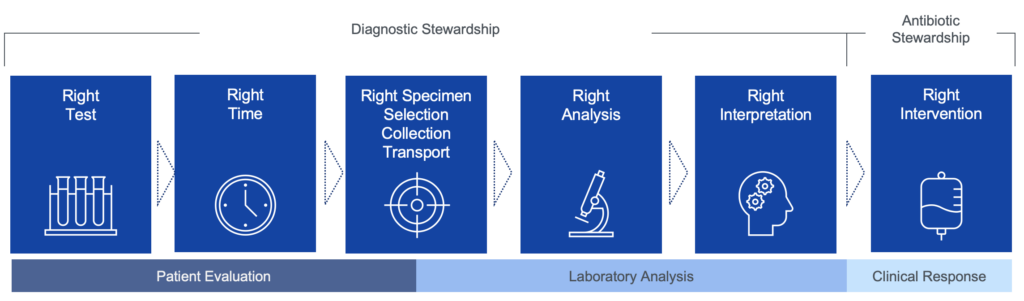
Adapted from Miller, JM er al. Clinical Infectious Diseases, 2024; 104.
As blood culture bottle supplies stabilize and clinicians return to evidence-based, recommended best practices, hospitals should track and reduce single-set blood culture utilization. For laboratories that receive a single-set blood culture, the CDC recommends alerting the clinician that the sample may result in inaccurate findings.12 The CDC recommends calculating the Blood Culture Contamination (BCC) rate monthly, using at least two blood culture sets collected within a 24-hour period.13
By avoiding single-set blood cultures and BCCs, hospitals can reduce the risk of misdiagnoses from false positive or false negative events, improve mitigation strategies, and improve the bottom line.
Evidence-Based Practices for Reducing Blood Culture Contamination (BCC)
Blood culture collection procedures should be regularly reviewed, and only trained staff should be collecting blood cultures.5 A summary of evidence-based best practices is provided below.14
1. Patient Selection
- Limit blood cultures to patients with a high probability of bacteremia.
- Avoid routine repeat cultures for Gram-negative bacteremia unless clinically indicated.
2. Skin Antisepsis
- Use alcohol-based disinfectants, such as 2% chlorhexidine with 70% isopropyl alcohol, to cleanse the venipuncture site.
- Allow the antiseptic to dry completely (≥30 seconds) before needle insertion.
3. Blood Culture Bottle Disinfection
- Disinfect the rubber septum of blood culture bottles with 70% isopropyl alcohol before inoculating samples.
- Avoid iodine-based disinfectants, which can degrade the rubber septum.
4. Blood Culture Collection Site & Technique
- Prefer peripheral venipuncture over central line draws whenever possible.
- Avoid drawing blood through intravascular catheters unless assessing catheter-related bloodstream infections.
- Use single-needle direct inoculation rather than the outdated double-needle technique.
5. Hand Hygiene and Sterile Gloves
- Perform proper hand hygiene before drawing blood.
- Use sterile gloves, especially if re-palpating the site after disinfection.
6. Blood Sampling and Volume
- Collect an adequate blood volume (20 mL per venipuncture).
- Use two separate venipunctures to reduce false positives.
- Avoid overfilling or underfilling bottles, which can increase contamination risk.
7. Phlebotomy Teams and Training
- Assign dedicated, trained phlebotomy teams to perform blood draws.
- Provide ongoing education and feedback to personnel to maintain best practices.
8. Surveillance and Feedback
- Monitor institutional BCC rates and provide individual feedback to phlebotomists.
- Implement real-time reporting of contaminated cultures to improve performance.
9. Initial Specimen Diversion Devices (ISDDs)
- Use diversion devices to discard the first portion of blood, which may contain skin contaminants.
- These devices have been shown to reduce BCC by up to 90% in clinical studies.
10. Multidisciplinary Quality Improvement Initiatives
- Engage infection control, microbiology, nursing, and laboratory teams in a coordinated effort to reduce contamination.
- Implement institution-wide performance improvement programs.
Key Takeaways
- With the recovery of blood culture bottle supply, hospitals should return to evidence-based guidelines and recommended practices for blood culture collection.
- Drawing at least two sets of blood cultures is critical to patient safety, diagnostic accuracy, and guideline compliance.
- Failing to follow proper blood culture collection practices increases the risk of false-positive and false-negative results, and can lead to the administration of unnecessary and/or inappropriate antibiotics.
- Hospitals should reassess current blood culture collection protocols and consider reminding staff on proper blood culture practices.
- Follow CDC recommendations on tracking and reporting blood culture contaminations (BCC) to drive continuous and targeted practice improvement.
Further Resources: CDC & Industry Guidelines
CDC Links:
- Prevent Adult Blood Culture Contamination
- Blood Culture Contamination: An Overview for Infection Control and Antibiotic Stewardship Programs Working with the Clinical Laboratory
- The Adult Blood Culture Contamination National Patient Safety Measure
American Society for Microbiology:
- Practical Guidance for Clinical Microbiology Laboratories: A Comprehensive Update on the Problem of Blood Culture Contamination and a Discussion of Methods for Addressing the Problem
- Laboratory Approaches to Determining Blood Culture Contamination Rates: An ASM Laboratory Practices Subcommittee report
BD BACTEC Statement:
- BD BACTEC Blood Culture Vial Supply
- BD BACTEC Hotline: 1.888.834.4332
Sources:
-
- https://www.idsociety.org/globalassets/idsa/multimedia/clinician-call-slides–qa/7.23.2024-qa.pdf
- https://bdbactec-update.com/wp-content/uploads/2024/12/BDBACTEC-Blood-Culture-Media-SupplyUpdate-December2024.pdf
- Lamy B, Dargere S, Arendrup MC, Parienti J-J, Tattevin P. How to optimize the use of blood cultures for the diagnosis of bloodstream infections? A state-of-the art. Front Microbiol. 2016;7:697.
- Bouza E, Sousa D, Rodríguez-Créixems M, Lechuz JG, Muñoz P. Is the volume of blood cultured still a significant factor in the diagnosis of bloodstream infections? J Clin Microbiol. 2007;45(9):2765-9.
- Centers for Disease Control and Prevention (CDC). Blood Culture Contamination: An Overview for Infection Control and Antibiotic Stewardship Programs Working with the Clinical Laboratory. Atlanta (GA): CDC. 2022. https://www.cdc.gov/antibiotic-use/core-elements/pdfs/fs-bloodculture-508.pdf
- Carrico R, Garrett H. Best Practices for Ensuring Clinical Continuity of Blood Cultures During Challenging Supply Chain Disruptions [PowerPoint Presentation]. Presented July 17, 2024. https://shea-online.org/wp-content/uploads/2024/08/SHEA-Town-Hall-Blood-Culture-Best-Practices.pdf
- Bunn, JD. Diagnostic Excellence: A New Quality Tool to Prevent Blood Culture Contamination [PowerPoint Presentation]. Presented December 13, 2023. https://reach.cdc.gov/event/diagnostic-excellence-new-quality-tool-prevent-blood-culture-contamination
- Bunn, JD. The Adult Blood Culture Contamination National Patient Safety Measure [PowerPoint Presentation]. November 1, 2023. https://www.cdc.gov/cliac/docs/november-2023/15_BCC.pdf
- Bunn JD, Cornish NE. Blood culture contamination and diagnostic stewardship: from a clinical laboratory quality monitor to a national patient safety measure. J Appl Lab Med. 2025;10(1):162-170.
- Palavecino EL, Campodónico VL, She RC. Laboratory approaches to determining blood culture contamination rates: an ASM Laboratory Practices Subcommittee report. J Clin Microbiol. 2024;62(2):e0102823.
- Clinical and Laboratory Standards Institute (CLSI). Principles and Procedures for Blood Cultures. 2nd ed. CLSI guideline M47. CLSI; 2022.
- https://www.cdc.gov/lab-quality/php/prevent-adult-blood-culture-contamination/sub-measure-single-set.html
- https://www.cdc.gov/lab-quality/php/prevent-adult-blood-culture-contamination/primary-measure.html
- Doern GV, Carroll KC, Diekema DJ, Garey KW, Rupp ME, Weinstein MP, Sexton DJ. A comprehensive update on the problem of blood culture contamination and a discussion of methods for addressing the problem. Clin Microbiol Rev. 2020 Jan;33(1):e00009-19. doi: 10.1128/CMR.00009-19